|
12/16/2023 0 Comments Modern pathology
Response to chemotherapy was assessed from local pathology reports for pathological complete response in breast and axillary lymph nodes. Eight hundred women with HER2-negative early invasive breast cancer were recruited. We compare data from central pathology review with report review and also the reporting behavior of the two central pathologists. Calculation of residual cancer burden was a simple and reproducible method of quantifying response to neoadjuvant chemotherapy as demonstrated by performance comparison of the two pathologists.Modern Pathology advance online publication, doi:10.1038/modpathol.2017.30.Ībstract = "The ARTemis Trial tested standard neoadjuvant chemotherapy±bevacizumab in the treatment of HER2-negative early breast cancer. In the ARTemis Trial central pathology review added little in the determination of pathological complete response but had a role in evaluating low levels of residual disease. For pathological complete response, report review was as good as central pathology review but for minimal residual disease, report review overestimated the extent of residual disease. Similar results were obtained for the between-treatment arm comparison both from the report review and the central pathology review. Good concordance was found between the two pathologists for residual cancer burden classes within the 65-patient quality assurance exercise (kappa 0.63 (95% CI: 0.57-0.69)). Residual cancer burden calculations were possible in 587/681 (86%) of the centrally reviewed patients, as 94/681 (14%) had positive sentinel nodes removed before neoadjuvant chemotherapy invalidating residual cancer burden scoring. 
Four hundred and eighty-three (71%) were assessed by JSJT, and 198 (29%) were assessed by EP. Full sample retrieval was obtained for 681 of the 781 patients (87%) who underwent surgery within the trial and were evaluable for pathological complete response. 10% of cases were double-reported to compare the central pathologists' reporting behavior. Pathologists recorded response to chemotherapy descriptively and also calculated residual cancer burden. Sections from the original core biopsy and surgical excision were centrally reviewed by one of two trial pathologists blinded to the local pathology reports. The centre operates on weekdays from 8:00-16:00.The ARTemis Trial tested standard neoadjuvant chemotherapy±bevacizumab in the treatment of HER2-negative early breast cancer. In addition, immunological or molecular studies can be conducted additionally at the cellular level. The basis of cytology services is to assess the morphology of cells taken using various methods from a disease site. Depending on their source material (cytological preparations or tissue sections), they are termed immunohistochemical or immunocytochemical stains. Other staining methods based on immunochemistry are in widespread use these are based on a reaction between an antigen and antibody. The cornerstone of pathology services is morphological and histological studies, centring on dried and paraffin-embedded, microtome-sliced and slide-mounted ultrathin tissue sections, which are stained using either routine or special staining methods. In addition, our centre conducts pathoanatomical autopsies mainly to evaluate treatment quality. These are the basis for classification of neoplasms and assessing residual disease or monitoring the therapeutic effects based on the requirements set forth in the WHO classification. 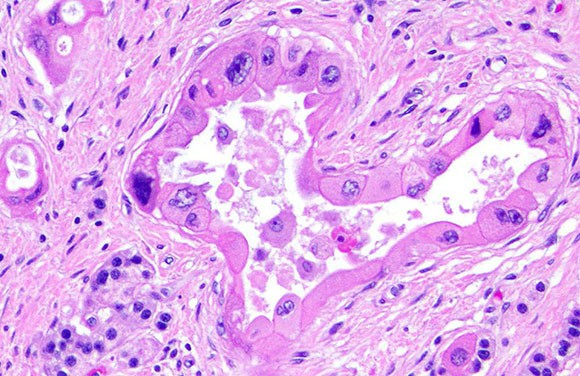
Diagnostics are based on morphological, immunological, molecular biology or cytogenetic investigative methods. Modern pathology is the study of diseases on the cellular and tissue level. Part of the Diagnostics Division, the Pathology Department is one of the leading diagnostics service units, covering the modern diagnostic needs of both general as well as specialized pathology.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
